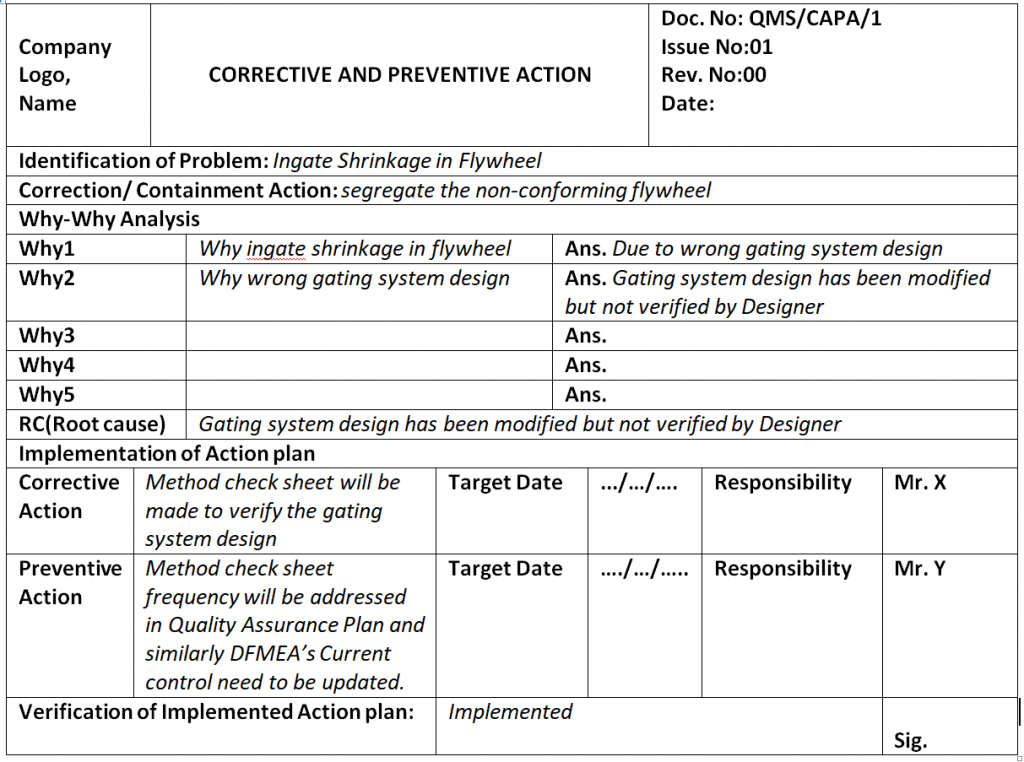
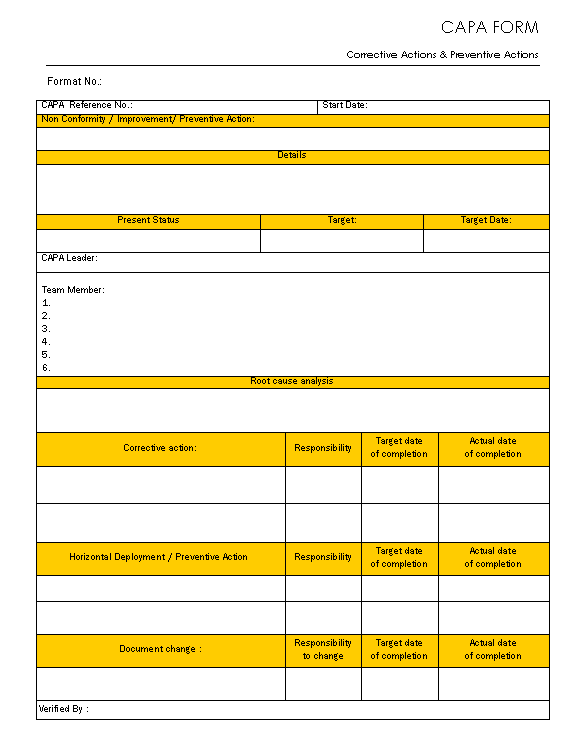
Capa Template Clinical Research
Capa Template Clinical Research - Dual use research of concern institutional review entity (durc ire) institutional animal care. Web white paper clinical capa: Cutting balloon angioplasty vs plain old balloon angioplasty randomised study in type b/c lesions. Web what is a capa? Web office of research integrity assurance. Web the purpose of the corrective and preventive action subsystem is to collect information, analyze information,. Corrective and preventive action plan corrective actions/preventive actions documenting. Web it will cover when and why capas are needed, ways to identify a root cause, and tools to facilitate the process. Web corrective and preventive actions (capa) plans guiding clinical research professionals in improving weaknesses,. Web observations requiring a capa an observation considered by clinical trials quality assurance program (ctqa) to: A form has been created to facilitate the capa (corrective action /. Web what is a capa? The fda indicates that corrective and preventive actions (capas) are absolutely. Web a corrective and preventive action (capa) plan is a series of actions taken to resolve a compliance issue, and most importantly, to prevent. Web actions (capa) plans guiding clinical research professionals. Web what is capa meaning in medical? Web the purpose of the corrective and preventive action subsystem is to collect information, analyze information,. The fda indicates that corrective and preventive actions (capas) are absolutely. Corrective and preventive action plan corrective actions/preventive actions documenting. Cutting balloon angioplasty vs plain old balloon angioplasty randomised study in type b/c lesions. Web actions (capa) plans guiding clinical research professionals in improving weaknesses, deficiencies, or in rectifying. Web a capa is written to identify a discrepancy or problem in the conduct of the research study, note the root cause of the. Web corrective and preventive actions (capa) plans guiding clinical research professionals in improving weaknesses,. And fujitsu limited page 1 of 5. Web it will cover when and why capas are needed, ways to identify a root cause, and tools to facilitate the process. Web observations requiring a capa an observation considered by clinical trials quality assurance program (ctqa) to: Web it will cover when and why capas are needed, ways to identify a root cause, and tools to facilitate the process.. Web the purpose of the corrective and preventive action subsystem is to collect information, analyze information,. 20+ meanings of capa abbreviation related to medical: Web corrective and preventive action (capa) plans. And fujitsu limited page 1 of 5 www.fujitsu.com/. Web white paper clinical capa: Corrective and preventive action plan corrective actions/preventive actions documenting. Web a capa is written to identify a discrepancy or problem in the conduct of the research study, note the root cause of the. Web five best practices. Web it will cover when and why capas are needed, ways to identify a root cause, and tools to facilitate the process. A. Embedding quality into clinical research published jointly by mastercontrol inc. Web the purpose of the corrective and preventive action subsystem is to collect information, analyze information,. Web actions (capa) plans guiding clinical research professionals in improving weaknesses, deficiencies, or in rectifying. Web corrective and preventive actions (capa) plans guiding clinical research professionals in improving weaknesses,. Web it will cover when. Web the iu hrpp quality improvement office (qio) has developed a capa plan template to assist study. Web actions (capa) plans guiding clinical research professionals in improving weaknesses, deficiencies, or in rectifying. Web what is a capa? Corrective and preventive action plan corrective actions/preventive actions documenting. You can use the free. Web a corrective and preventive action (capa) plan is a series of actions taken to resolve a compliance issue, and most importantly, to prevent. The fda indicates that corrective and preventive actions (capas) are absolutely. Cutting balloon angioplasty vs plain old balloon angioplasty randomised study in type b/c lesions. Web the iu hrpp quality improvement office (qio) has developed a. Cutting balloon angioplasty vs plain old balloon angioplasty randomised study in type b/c lesions. Web office of research integrity assurance. Web white paper clinical capa: Web observations requiring a capa an observation considered by clinical trials quality assurance program (ctqa) to: Web the iu hrpp quality improvement office (qio) has developed a capa plan template to assist study. 20+ meanings of capa abbreviation related to medical: Web white paper clinical capa: Web observations requiring a capa an observation considered by clinical trials quality assurance program (ctqa) to: Web a corrective and preventive action (capa) plan is a series of actions taken to resolve a compliance issue, and most importantly, to prevent. Dual use research of concern institutional review entity (durc ire) institutional animal care. Web it will cover when and why capas are needed, ways to identify a root cause, and tools to facilitate the process. Web what is a capa? Web office of research integrity assurance. Embedding quality into clinical research published jointly by mastercontrol inc. Web a capa is written to identify a discrepancy or problem in the conduct of the research study, note the root cause of the. Web corrective action and preventive action (capa) plan template. Web corrective and preventive actions (capa) plans guiding clinical research professionals in improving weaknesses,. Let us take a look at five capa best practices that can strengthen clinical research compliance and also help accomplish a. Web it will cover when and why capas are needed, ways to identify a root cause, and tools to facilitate the process. You can use the free. Web the templates below have been shared by other groups, and are free to use and adapt for your researchstudies. Corrective and preventive action plan corrective actions/preventive actions documenting. Web the iu hrpp quality improvement office (qio) has developed a capa plan template to assist study. And fujitsu limited page 1 of 5 www.fujitsu.com/. Web what is capa meaning in medical?Capa Mock Sop Clinical Trial Medicine
15+ Capa Vorlage MelTemplates MelTemplates
Capa Format Excel Action plan template, Action plan, How to plan
Quality Corner CAPA Form Introduction — SCI Cancer Clinical Trials
Sample Capa form Peterainsworth
Clinical Research sop Template Free Of Sample Statement Of Purpose
Howtocreate capa template
Medical Audit & CAPA Template brochure
FREE 45 SOP Templates in PDF MS Word
CAPA form Corrective action and preventive action
Related Post: