Capa Template Fda
Capa Template Fda - Web corrective and preventive action plan (capa) • a system for resolving quality issues • resolve/correct problem and keep it from. Application form is downloaded from www.fda.gov.ph. If you’ve ever had a workplace. Determine if the firm manufactures or imports a tracked device. The integrated application form in xls or xlsx format is. 1) creating and submitting a request; Web as noted throughout this guide, capa is an important process for your medical device company. Web capa refers to corrective and preventative actions. Food and drug administration (fda) the fda’s quality system regulations, 21 cfr 820.100. Our free capa form template has all the required fields and is a quick, readymade solution. Web corrective and preventive actions (capa) form template. Web what is capa per ich q10? Web capa refers to corrective and preventative actions. Web complete capa format in excel download in just several minutes by simply following the guidelines listed below: Web get started with this customizable capa form template. Web what is capa per ich q10? Web capa fda is a quality management strategy used in the manufacturing and production industries to meet the intent of the fda 21 cfr 820.100. Food and drug administration (fda) the fda’s quality system regulations, 21 cfr 820.100. Web capa procedures • your firm failed to establish, maintain, and implement a corrective and. Web it discusses capa within iso 9001 and within the regulation fda 21 cfr 820 and outlines how and what data. Web complete capa format in excel download in just several minutes by simply following the guidelines listed below: 12 april 2016 form no. Added use of qmis form. Determine if the firm manufactures or imports a tracked device. Web corrective and preventive action plan (capa) • a system for resolving quality issues • resolve/correct problem and keep it from. (corrective and preventive actions) structured approach to the investigation process should be used. Web corrective and preventive actions (capa) form template. Web complete capa format in excel download in just several minutes by simply following the guidelines listed below:. If you’ve ever had a workplace. Our free capa form template has all the required fields and is a quick, readymade solution. Web complete capa format in excel download in just several minutes by simply following the guidelines listed below: Web what is capa per ich q10? Web corrective action and preventive action plan date effective: Web a corrective and preventive action (capa) system is a roadmap of processes regulators expect. Web it discusses capa within iso 9001 and within the regulation fda 21 cfr 820 and outlines how and what data. 12 april 2016 form no. Food and drug administration (fda) the fda’s quality system regulations, 21 cfr 820.100. Web medical device tracking. Web what is capa per ich q10? Web a corrective and preventive action (capa) system is a roadmap of processes regulators expect. Web capa fda is a quality management strategy used in the manufacturing and production industries to meet the intent of the fda 21 cfr 820.100. Web it discusses capa within iso 9001 and within the regulation fda 21. Web it discusses capa within iso 9001 and within the regulation fda 21 cfr 820 and outlines how and what data. Web corrective and preventive actions (capa) form template. Added ora capa procedure to reference section. Web capa fda is a quality management strategy used in the manufacturing and production industries to meet the intent of the fda 21 cfr. Determine if the firm manufactures or imports a tracked device. Web medical device tracking. Web as noted throughout this guide, capa is an important process for your medical device company. Web a corrective and preventive action (capa) system is a roadmap of processes regulators expect. Web it discusses capa within iso 9001 and within the regulation fda 21 cfr 820. The guide to capa & root. Our free capa form template has all the required fields and is a quick, readymade solution. Web it’s stressful to receive an observation or warning letter from the food and drug administration for corrective and. Web what is capa per ich q10? Capa is a concept within good manufacturing practices (gmp). Application form is downloaded from www.fda.gov.ph. Food and drug administration (fda) the fda’s quality system regulations, 21 cfr 820.100. Our free capa form template has all the required fields and is a quick, readymade solution. Web corrective and preventive action plan (capa) • a system for resolving quality issues • resolve/correct problem and keep it from. The integrated application form in xls or xlsx format is. Web a corrective and preventive action (capa) system is a roadmap of processes regulators expect. Web corrective and preventive actions (capa) form template. 1) creating and submitting a request; Added use of qmis form. Web get started with this customizable capa form template. Web as noted throughout this guide, capa is an important process for your medical device company. Web capa procedures • your firm failed to establish, maintain, and implement a corrective and preventive action procedure, as required by 820.100(a). Capa is a concept within good manufacturing practices (gmp). Added ora capa procedure to reference section. If you’ve ever had a workplace. (corrective and preventive actions) structured approach to the investigation process should be used. Web capa fda is a quality management strategy used in the manufacturing and production industries to meet the intent of the fda 21 cfr 820.100. Web capa refers to corrective and preventative actions. The guide to capa & root. Web complete capa format in excel download in just several minutes by simply following the guidelines listed below:CAPA Solutions Document Control CAPA Management Software
SOP For Corrective Action and Preventive Actions Pharmaceutical
LOGO
Medical Audit & CAPA Template brochure
Corrective and Preventive Action Format CAPA with Example Download
What FDA Expects to See as Part of Your CAPA Process Free Download
LOGO
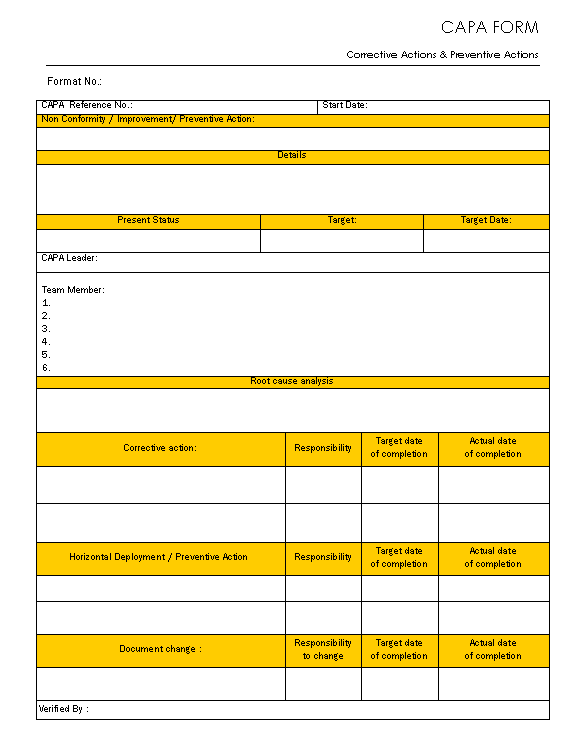
CAPA form Corrective action and preventive action
Customs & International Trade Law Expert483 Inspection Observation
Corrective and preventive action plan CAPA report form
Related Post: