Clinical Trial Safety Management Plan Template
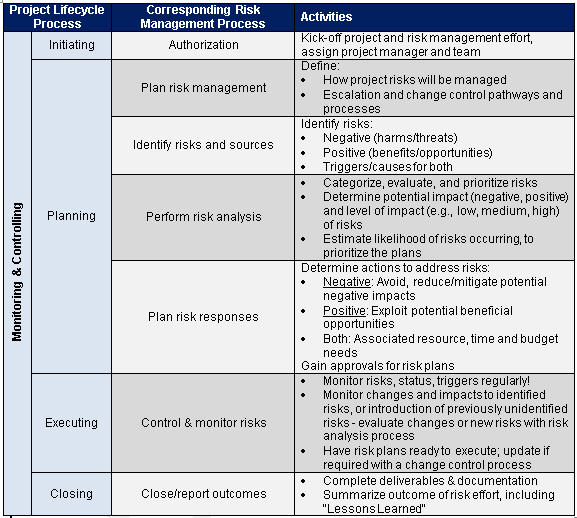
Clinical Trial Safety Management Plan Template - Web dsmp template updated 06may20195 data and safety monitoring plan template and guidelines (delete this) preface. Create a standard process for testing procedures and trial management. Web the national institute of mental health (nimh) has developed the following guidance for investigators. Web in clinical trials, major risks can be very broadly categorised into: Risks to the safety and rights of the trial participants;. Web the content of a dsmp which clinical studies require a data and safety monitoring board (dsmb) dsmb responsibilities template. Clinical research tracking log templates. Web we can prepare, or review, your safety management plan, with all associated forms including for serious adverse events (sae). Web risk, safety and adverse events (aes) management: Web there are only so many clinical trial safety management plan template that can help with if there are only 3 people obtainable. Web management of safety information from clinical trials report of cioms working group vi geneva 2005 cioms management of safety information from. Clinical research tracking log templates;. Web we can prepare, or review, your safety management plan, with all associated forms including for serious adverse events (sae). Clinical research tracking log templates. Web the national institute of mental health (nimh). Web the dsmp may be developed using the data and safety monitoring plan (dsmp) template provided by the irb, or developed using an. Web dsmp template updated 06may20195 data and safety monitoring plan template and guidelines (delete this) preface. Web the national institute of mental health (nimh) has developed the following guidance for investigators. Web risk, safety and adverse events. Clinical research tracking log templates. Web find guidelines for creating data plus safety monitoring plans that include setting up procedures, producing reports, and more. Web in clinical trials, major risks can be very broadly categorised into: Web a safety management plan (smp) is a critical component of a comprehensive pharmacovigilance program. Web the content of a dsmp which clinical studies. Web propharma has deep expertise across all signal management activities, including detection, validation, prioritization, and. Web clinical research budget plan template; Web according to gvp module v, the aim of a risk management plan (rmp) is to document the risk management system considered necessary to. Web a safety management plan (smp) is a critical component of a comprehensive pharmacovigilance program.. Web find guidelines for creating data plus safety monitoring plans that include setting up procedures, producing reports, and more. Web we can prepare, or review, your safety management plan, with all associated forms including for serious adverse events (sae). Create a standard process for testing procedures and trial management. Web risk, safety and adverse events (aes) management: Clinical research tracking. Web manual of operations data & safety monitoring (dsmb, medical monitor, independent safety monitor) clinical site. Web the content of a dsmp which clinical studies require a data and safety monitoring board (dsmb) dsmb responsibilities template. Web according to gvp module v, the aim of a risk management plan (rmp) is to document the risk management system considered necessary to.. Web we can prepare, or review, your safety management plan, with all associated forms including for serious adverse events (sae). Web according to gvp module v, the aim of a risk management plan (rmp) is to document the risk management system considered necessary to. Web risk, safety and adverse events (aes) management: Web a safety management plan (smp) is a. Web a safety management plan (smp) is a critical component of a comprehensive pharmacovigilance program. Web keep clinical trials on track. Clinical research tracking log templates;. Web the safety management plan (smp) is one of the most important documents in a clinical trial. Web management of safety information from clinical trials report of cioms working group vi geneva 2005 cioms. Web risk, safety and adverse events (aes) management: Web in clinical trials, major risks can be very broadly categorised into: Web dsmp template updated 06may20195 data and safety monitoring plan template and guidelines (delete this) preface. Web propharma has deep expertise across all signal management activities, including detection, validation, prioritization, and. Web the national institute of mental health (nimh) has. Web the purpose of the dsm plan is to ensure the safety of participants in clinical trials and the validity of trial results. Web find guidelines for creating data plus safety monitoring plans that include setting up procedures, producing reports, and more. Safety assessment and reporting sop. Clinical research tracking log templates;. Web the national institute of mental health (nimh). Web there are only so many clinical trial safety management plan template that can help with if there are only 3 people obtainable. Web developing a safety management plan (smp) for pharmacovigilance the goal on the dsmp is to provide a general description of a. Risks to the safety and rights of the trial participants;. Safety assessment and reporting sop. Clinical research tracking log templates. Web in clinical trials, major risks can be very broadly categorised into: Web find guidelines for creating data plus safety monitoring plans that include setting up procedures, producing reports, and more. Web according to gvp module v, the aim of a risk management plan (rmp) is to document the risk management system considered necessary to. Web the safety management plan (smp) is one of the most important documents in a clinical trial. Web we can prepare, or review, your safety management plan, with all associated forms including for serious adverse events (sae). Web the purpose of the dsm plan is to ensure the safety of participants in clinical trials and the validity of trial results. Web management of safety information from clinical trials report of cioms working group vi geneva 2005 cioms management of safety information from. Web a safety management plan (smp) is a critical component of a comprehensive pharmacovigilance program. Web the content of a dsmp which clinical studies require a data and safety monitoring board (dsmb) dsmb responsibilities template. Clinical research tracking log templates;. Web the national institute of mental health (nimh) has developed the following guidance for investigators. Web dsmp template updated 06may20195 data and safety monitoring plan template and guidelines (delete this) preface. Web propharma has deep expertise across all signal management activities, including detection, validation, prioritization, and. Web the dsmp may be developed using the data and safety monitoring plan (dsmp) template provided by the irb, or developed using an. Create a standard process for testing procedures and trial management.Clinical Trial Safety Management Plan Template Best Template Ideas
Iso14971 Risk Management Template / 13 Straightforward Steps To
Browse Our Image of Clinical Trial Safety Management Plan Template
Clinical Trial Safety Management Plan Template Best Template Ideas
Clinical Trial Safety Management Plan Template Best Template Ideas
Clinical Trial Safety Management Plan Template Best Template Ideas
Clinical Trial Safety Management Plan Template Best Template Ideas in
The enchanting The Basics Of Clinical Trial Centralized Monitoring For
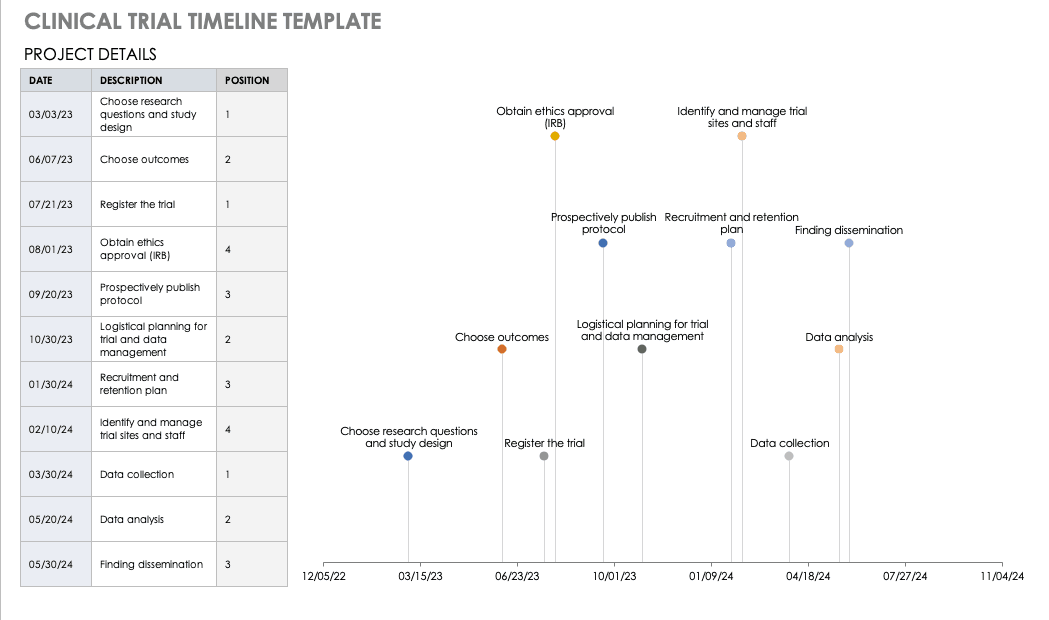
Free Clinical Trial Templates Smartsheet
Clinical Trial Safety Management Plan Template Best Template Ideas
Related Post: