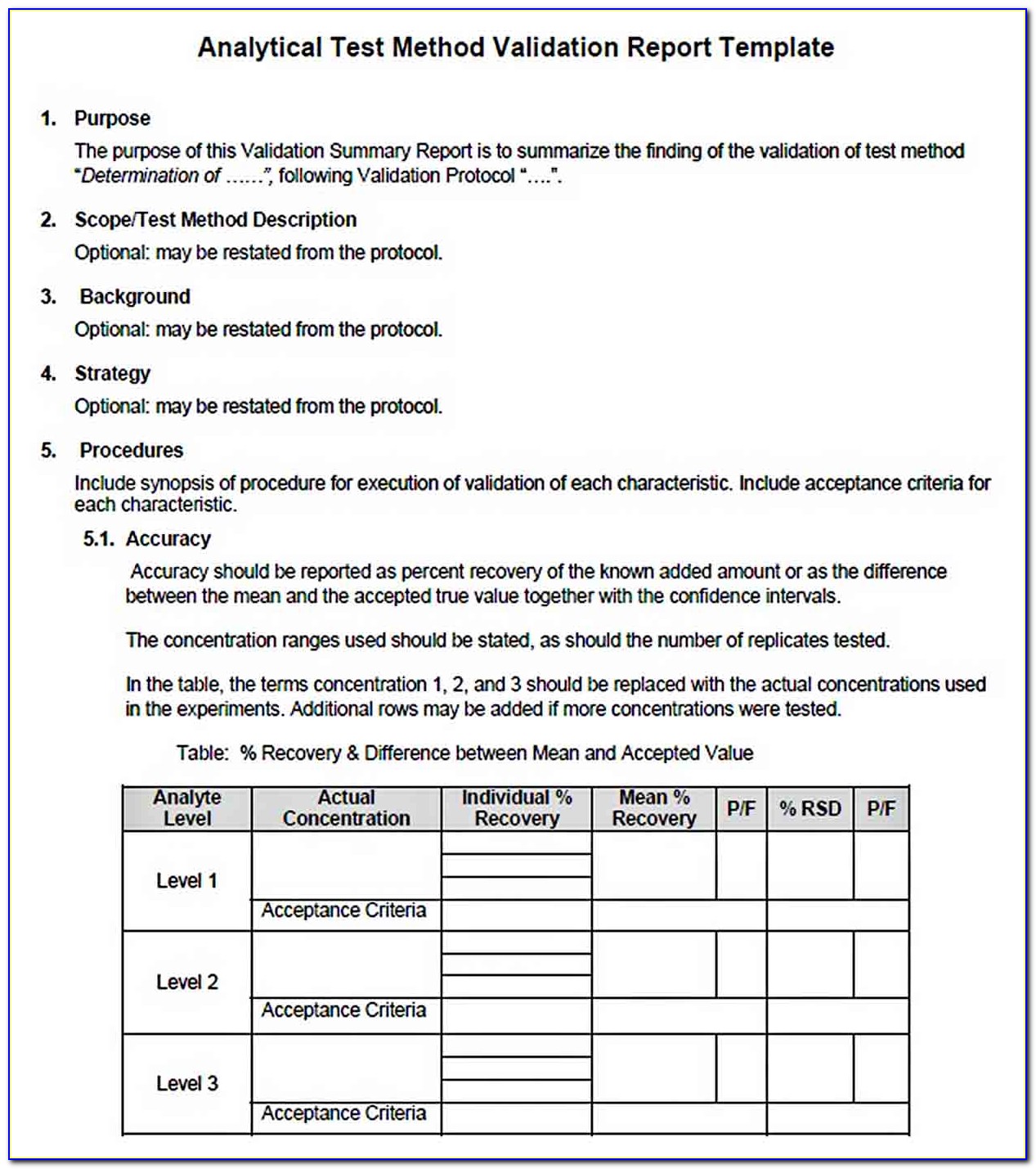
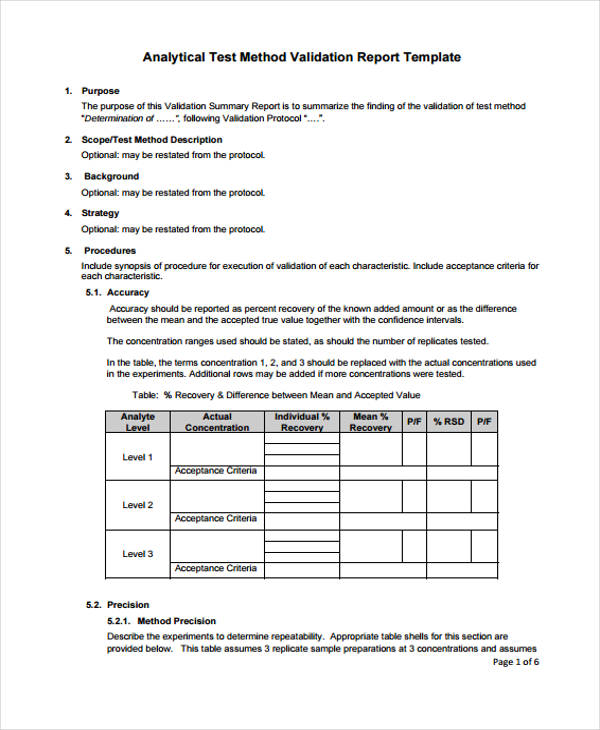
Validation Protocol Template
Validation Protocol Template - Web at step 2 of the ich process, a consensus draft text or guideline, agreed by the appropriate ich expert working group, is. Cleaning validation is a procedure of establishing evidence that. These are the abbreviations we use in the medical device industry for the three steps of. Web an equipment validation protocol is a written plan stating how equipment qualification will be conducted. It details factors such as product characteristics, production equipment, test scripts and methods, test parameters and acceptance criteria, test checksheets and final approval. However, validation plans, experiments, results and. Templates repository for software development process (click here) or. Web 1 objective the objective of this protocol is to define the procedure used and the acceptance criteria for validation of [insert full product description (eg. Web validation strategy this process validation will consist of three multi vitamin tablet lots of commercial size (xxxxkg). Web tance criteria for making conclusions. Web develop working processes with a process validation protocol template. Web what is iq, oq, pq? Templates repository for software development process (click here) or. Web here are the details of validation protocol & report format + types pdf ppt. Web this verification is achieved by subjecting the equipment to a range of fully documented inspections and tests. Validation studies, stability studies, and clinical studies are examples of written. Web clia rules and guidance3 are silent on minimum requirements for validation protocols; Web this equipment validation protocol template is designed to ensure that the equipment in question is safe, effective, and compliant. Web this verification is achieved by subjecting the equipment to a range of fully documented inspections. Web what is iq, oq, pq? Web this verification is achieved by subjecting the equipment to a range of fully documented inspections and tests. Analytical validation seeks to demonstrate that the. Study this protocol was generated and approved to. Web an equipment validation protocol is a written plan stating how equipment qualification will be conducted. Web use this process validation report template in the pharmaceutical industry to document everything properly. Web this verification is achieved by subjecting the equipment to a range of fully documented inspections and tests. Web develop working processes with a process validation protocol template. However, validation plans, experiments, results and. Analytical validation seeks to demonstrate that the. Web sop for template for process validation protocol in pharma. Web at step 2 of the ich process, a consensus draft text or guideline, agreed by the appropriate ich expert working group, is. Web this verification is achieved by subjecting the equipment to a range of fully documented inspections and tests. Web published 4 may 2023 what is cleaning validation?. Web this verification is achieved by subjecting the equipment to a range of fully documented inspections and tests. Cleaning validation is a procedure of establishing evidence that. However, validation plans, experiments, results and. Analytical validation seeks to demonstrate that the. Web template for an example methods validation protocol 171 i. Web this equipment validation protocol template is designed to ensure that the equipment in question is safe, effective, and compliant. Web published 4 may 2023 what is cleaning validation? It details factors such as product characteristics, production equipment, test scripts and methods, test parameters and acceptance criteria, test checksheets and final approval. Study this protocol was generated and approved to.. Web develop working processes with a process validation protocol template. Web published 4 may 2023 what is cleaning validation? Web template for an example methods validation protocol 171 i. Cleaning validation is a procedure of establishing evidence that. Web pdf template, an installation qualification template is used to complete the process validation protocol by properly. Sop for process validation is a. Define the facilities, systems, equipment, or processes in the scope of the. Web use this process validation report template in the pharmaceutical industry to document everything properly. Web develop working processes with a process validation protocol template. Web this equipment validation protocol template is designed to ensure that the equipment in question is safe,. Web validation strategy this process validation will consist of three multi vitamin tablet lots of commercial size (xxxxkg). Web sop for template for process validation protocol in pharma. Cleaning validation is a procedure of establishing evidence that. More templates to download on the: Analytical validation seeks to demonstrate that the. More templates to download on the: Web clia rules and guidance3 are silent on minimum requirements for validation protocols; Web published 4 may 2023 what is cleaning validation? Web pdf template, an installation qualification template is used to complete the process validation protocol by properly. Web use this process validation report template in the pharmaceutical industry to document everything properly. Define the facilities, systems, equipment, or processes in the scope of the. Study this protocol was generated and approved to. Web tance criteria for making conclusions. Web 1 objective the objective of this protocol is to define the procedure used and the acceptance criteria for validation of [insert full product description (eg. Web what is iq, oq, pq? Sop for process validation is a. However, validation plans, experiments, results and. Templates repository for software development process (click here) or. Web validation strategy this process validation will consist of three multi vitamin tablet lots of commercial size (xxxxkg). Web template for process validation protocol objective to provide documented evidence with high degree of. Analytical validation seeks to demonstrate that the. Web this verification is achieved by subjecting the equipment to a range of fully documented inspections and tests. Web an equipment validation protocol is a written plan stating how equipment qualification will be conducted. Web process validation protocol template or format for the products manufactured in the pharmaceutical product manufacturing. These are the abbreviations we use in the medical device industry for the three steps of.Analytical Test Method Validation Protocol Template
Validation Protocols Reports Procedure
Template for Process Validation Protocol Verification And Validation
Analytical Method Validation Protocol Sample
Software Validation Templates
Excel Spreadsheet Validation Protocol Template
TEM260_Cleaning_Validation_Protocol_Template_sample.pdf Chemistry
Tem290 Process Validation Protocol Template Sample Verification And
TEM280 Packaging Validation Protocol Template Sample Verification
process validation protocol DriverLayer Search Engine
Related Post: